Clinical
Greens' worldwide reputation in the production and global distribution of clinical trials materials comes from over 40 years of service to the industry. Our offices in England and USA enable us to manage and produce every aspect of your biggest and smallest trials. Just as importantly, we understand the issues facing you of making sure all your materials arrive on site on time ready to use.
We can provide printed materials for clinical trials, as well as marketing collateral to assist with patient recruitment.
Key services:
- CRFs (case report forms)
- Patient questionnaires
- Patient screening forms
- Patient diaries
- Brochures
- Letters
- ID cards
Site Binders:
The ISFs and pharmacy site binders are large and complicated documents and our approach is to be as flexible as possible, working with whichever format of files our clients supply, utilising our expertise and minimising any preparation work for our clients.
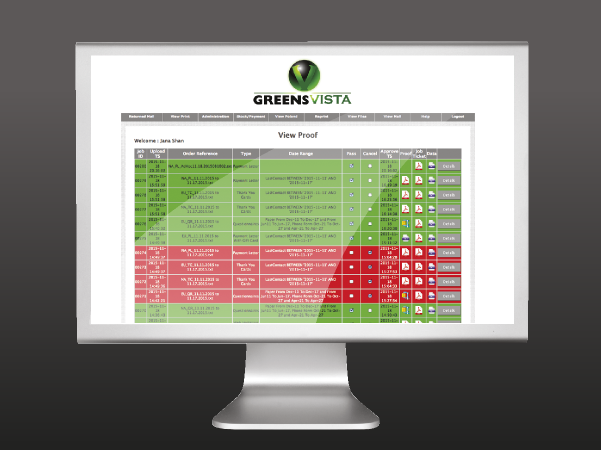
We have streamlined the pre-press set-up with eProofs and hard copy samples available within 48 hours for review, to reduce lead times, as we know SIV deadlines are so often tight and it's crucial the study files are at the site when the CRA needs them. We also offer a secure file upload facility to transfer the documents easily.
Greens work closely with our clients to find innovative and cost efficient solutions to problems. A US CRO utilised our logistics facility in the UK to store and distribute medical equipment used in the trial with the study files, which ensured no customs delays when shipping to Europe and a cost saving as we could minimise the number of shipments to sites. We have a fully secured warehousing facility with CCTV for the study material to be stored and distributed to site on demand via our bespoke web ordering facility that offers complete point-to-point track and trace of your deliveries.
We are also well versed in personalising the study binders for sites, with covers and spines detailing investigator names and site numbers, and any site-or language-specific documents filed within the binders. We have strict QC procedures in place, in accordance with our ISO 9001 accreditation, to ensure the integrity of the shipments being despatched to site.
Greens specialise in the production and distribution of investigator site files for clinical trials, shipping thousands of binders globally each month, both direct to site and to country offices.